 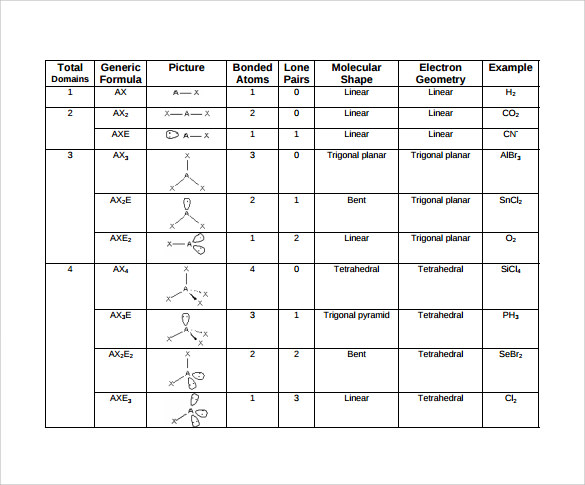
By contrast, nonbonding electrons move under the attractive influence of only one nucleus and thus spread out more in space. Bonding pairs are attracted by the two nuclei. The Effect of Nonbonding Electrons and Multiple Bonds on Bond Anglesīond angles decrease as the number of nonbonding electron pairs increases. Describe the molecular geometry in terms of the angular arrangement of the bonding pairsįour or Fewer Valence-Shell Electron PairsĪ double or triple bond has essentially the same effect on bond angles as a single bond and is therefore counted as one bonding pair (that is, one electron region) when predicting geometry.Count the total number of electron pairs around the central atom and arrange them in the way that minimizes electron-pair repulsions.Sketch the Lewis dot structure of the molecule or ion.How to predict molecular geometries using VSEPR model: 
However, according to molecular geometry it is a trigonal pyramidal because it has only 3 bonds. For example, NH 3 is a tetrahedral according to electron-pair geometry because it has 3 bonds and an unshared pair of electron. Molecular geometries only include BONDING pairs. When describing the shapes of molecules, we always give the molecular geometry rather than the electron-pair geometry. Molecular geometry: the arrangement in space of the atoms of a molecule.Electron-pair geometry: 3-D arrangement of electron pairs around an atom according to the VSEPR model.Two types of valence-shell electron pairs bonding pairs and nonbonding pairs.Electrons repel one another hence, the best arrangement of a given number of electron pairs is the one that minimizes the repulsions among them.Valence-shell electron-pair repulsion (VSEPR) model: model that accounts for the geometric arrangements of shared and unshared electron pairs around a central atom in terms of the repulsions between electron pairs.The Valence-Shell Electron Pair-Repulsion Model Instead, it is a counting procedure that accurately predicts the three-dimensional structures of a large number of compounds, which cannot be predicted using the Lewis electron-pair approach. The VSEPR model is not a theory it does not attempt to explain observations. The VSEPR model can predict the structure of nearly any molecule or polyatomic ion in which the central atom is a nonmetal, as well as the structures of many molecules and polyatomic ions with a central metal atom. VSEPR Model" data-cke-saved-href="/Bookshelves/General_Chemistry/Map:_Chemistry_-_The_Central_Science_(Brown_et_al.)/09:_Molecular_Geometry_and_Bonding_Theories/9.02:_The_VSEPR_Model" href="/Bookshelves/General_Chemistry/Map:_Chemistry_-_The_Central_Science_(Brown_et_al.)/09:_Molecular_Geometry_and_Bonding_Theories/9.02:_The_VSEPR_Model" data-quail-id="12">9.2: The VSEPR Model 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
